Most people assume supplements are supplements. You buy a bottle, trust the label, swallow the capsule. But the standard to which that capsule was manufactured determines almost everything: how pure the ingredient actually is, how much of it you absorb, and what else — wanted or not — came along for the ride.
The term pharmaceutical grade is not marketing language. It refers to a specific manufacturing classification with defined purity thresholds, testing protocols, and regulatory requirements. And the gap between it and the food-grade standard that governs most supplements on the market is wider than most consumers realise.
This article breaks down what pharmaceutical grade supplements actually means, how the four manufacturing tiers compare, what GMP certification does and does not guarantee, and why grade alone is only part of the picture.
What does "pharmaceutical grade" actually mean?
Pharmaceutical grade is the highest classification in supplement and ingredient manufacturing. To qualify, an ingredient or finished product must meet the purity and quality standards set by a recognised pharmacopoeia — typically the United States Pharmacopeia (USP), the British Pharmacopoeia (BP), or the European Pharmacopoeia (Ph.Eur.).
In practice, pharmaceutical grade requires:
- Purity of 99% or greater — the active ingredient makes up at least 99% of the substance, with tightly controlled limits on contaminants, fillers, and by-products
- Documented identity testing — every batch must be verified to contain exactly what it claims
- Limits on elemental impurities — heavy metals such as lead, arsenic, cadmium, and mercury must fall within pharmacopoeial thresholds
- Full batch documentation — traceability from raw material sourcing through to finished product
- Manufacturing in a GMP-certified pharmaceutical facility — not just a food-manufacturing or nutraceutical plant
These standards exist because pharmaceutical grade manufacturing was originally designed for prescription medications — contexts where purity and consistency are non-negotiable. Applying those same standards to nutritional supplements is a deliberate choice, not a legal requirement. And that distinction matters.
The four manufacturing grades explained
Not all supplement ingredients are made equal. Raw materials used in supplement production are classified into four tiers, each with different purity benchmarks and testing requirements.
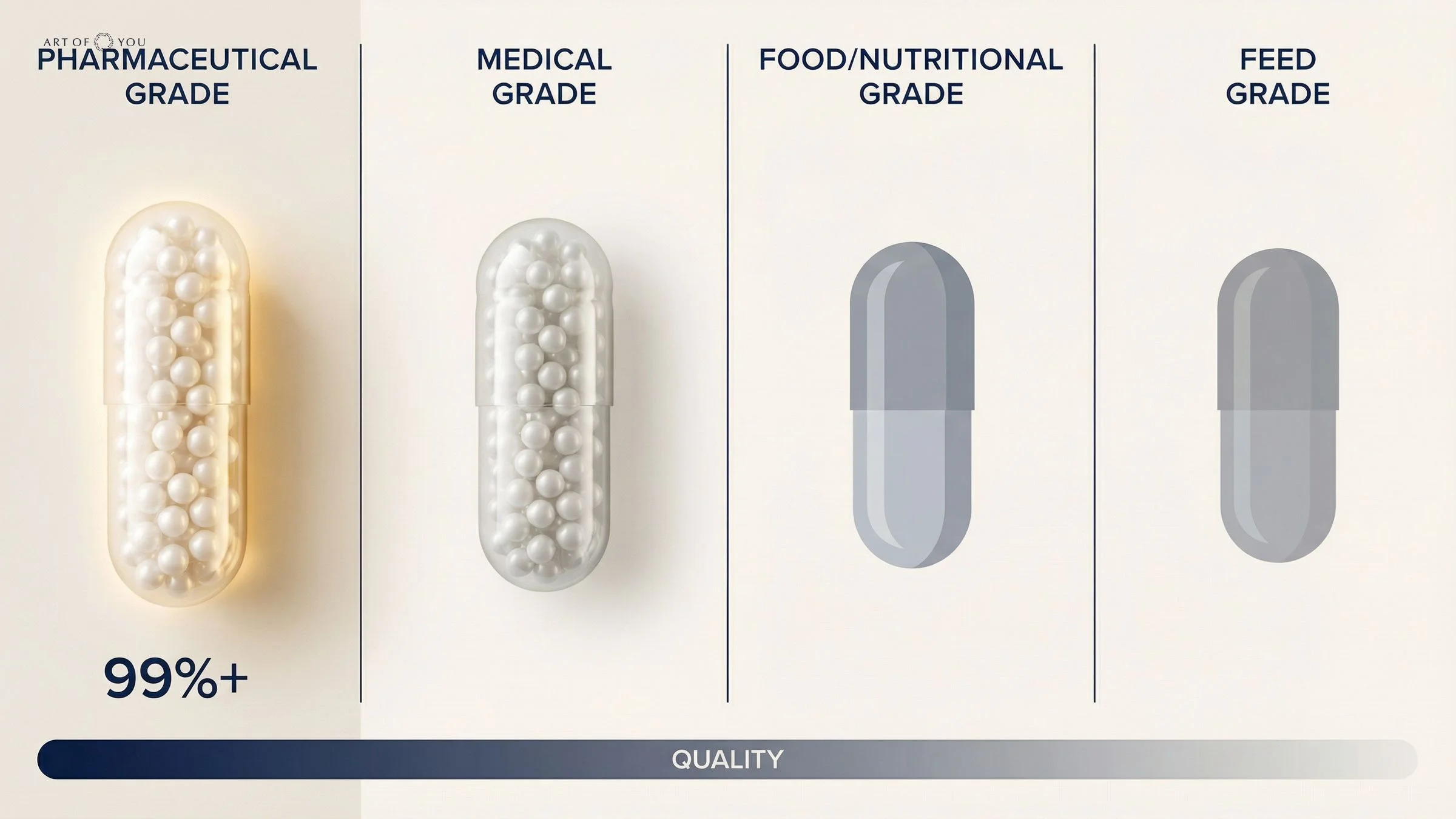
| Grade | Intended Use | Typical Purity | Testing Standard | Heavy Metal Limits |
|---|---|---|---|---|
| Pharmaceutical | Human prescription and OTC medicine | 99%+ | USP / BP / Ph.Eur. monographs | Strict pharmacopoeial limits |
| Medical | Medical devices, some diagnostics | 95–99% | ISO and pharmacopoeial standards | Controlled, product-specific |
| Food / Nutritional | Human food and most supplements | 80–95% | Food safety regulations (CODEX, FDA Food) | Broader tolerance ranges |
| Feed | Veterinary and agricultural use | Variable (lowest) | Agricultural standards | Minimal controls |
The purity gap between pharmaceutical and food grade is significant. A food-grade vitamin C ingredient, for example, may contain 80–95% of the labelled active compound — with the remainder being moisture, binders, processing residues, or unlisted compounds. A pharmaceutical-grade equivalent is verified at 99%+ with documented limits on every impurity.
For an ingredient like iron, where the exact dose matters for both efficacy and tolerability, that variance is not trivial.
Why most supplements are food grade (and what that costs you)
The global supplement industry is not regulated like the pharmaceutical industry. In the EU and the UK, food supplements are governed by food law, not medicines law — meaning manufacturers are not legally required to use pharmaceutical-grade raw materials. In the US, the FDA dietary supplement regulations (21 CFR Part 111) establish GMP requirements for manufacturing processes, but do not mandate pharmaceutical-grade ingredients.
The result is a market where the vast majority of supplements are manufactured using food-grade or nutritional-grade ingredients. This is not illegal. It is not necessarily dangerous. But it does mean:
- Ingredient purity can vary batch to batch
- Contaminant limits are less stringent
- Testing requirements are less rigorous than pharmaceutical standards
- The consumer has limited visibility into what is actually in the product
A 2024 analysis by BSCG (Banned Substances Control Group) found that even GMP-certified supplement facilities could produce products that fail quality testing — because GMP certification applies to the facility's processes, not to individual finished products.
This is the structural challenge with the supplement market: process compliance does not automatically equal product quality.
What GMP certification means — and what it does not
Good Manufacturing Practice (GMP) is a system of guidelines that governs how products are manufactured. In the supplement industry, GMP certification is awarded to facilities — not to individual products.
GMP certification (whether NSF International, NPA, or EU GMP) tells you:
- The manufacturing facility operates with documented quality systems
- Processes for cleanliness, personnel training, equipment calibration, and record-keeping are in place
- The facility undergoes independent audits
GMP certification does not guarantee:
- That a specific product passed contaminant testing
- That the raw materials used were pharmaceutical grade
- That every finished batch was independently verified
- That the label claims are accurate within tight tolerances
NSF International's product certification program (NSF/ANSI 173) goes further than facility GMP — testing individual products for label accuracy, 290+ banned substances, and contaminant thresholds. USP Verified adds another layer by confirming potency and purity of specific formulations. These are meaningful additional signals.
But even these third-party programs are voluntary, and most supplement brands do not pursue them.

What to look for: third-party testing and certificates of analysis
When evaluating a supplement's quality credentials, three things matter most:
1. Certificate of Analysis (CoA) A CoA is a batch-level document from an independent laboratory confirming the identity, purity, and potency of an ingredient or finished product. Reputable brands make CoAs available on request or on their website. If a brand cannot provide a CoA for a specific batch, that is a meaningful gap.
2. Heavy metal testing Pharmacopoeias set strict limits for lead, arsenic, cadmium, and mercury. Pharmaceutical-grade raw materials must meet these limits at the ingredient level before they ever enter a capsule. Food-grade ingredients operate under broader tolerances. Heavy metal testing results should be documented in the CoA.
3. Third-party verification Independent testing by organisations like NSF International, Informed Sport, or USP Verified provides verification that goes beyond manufacturer self-reporting. For athletes or individuals taking multiple supplements, third-party verified products provide an additional layer of assurance.
These are the practical signals that separate pharmaceutical-grade transparency from food-grade opacity.
Why grade alone is not enough: bioavailability and delivery form
Purity is necessary but not sufficient. How an ingredient is delivered to the body — its delivery form — determines how much of that pharmaceutical-grade compound your body actually absorbs and uses.
This is the concept of bioavailability: the proportion of a nutrient that enters circulation and is available for use. It varies dramatically by:
- The chemical form of the ingredient (e.g., methylfolate vs. folic acid; methylcobalamin vs. cyanocobalamin)
- The delivery architecture (standard compressed tablet vs. enteric-coated vs. multi-layered beadlet)
- The timing and environment of release in the digestive tract
- Nutrient interactions within a single capsule — some compounds compete for absorption pathways, some enhance each other
A pharmaceutical-grade ingredient delivered in a compressed tablet alongside competing compounds may absorb significantly less effectively than the same ingredient delivered as a precisely engineered individual beadlet.

Beadlet encapsulation: the delivery solution
The most advanced approach to this problem is individual beadlet encapsulation — where each ingredient is formed into its own micro-bead with a distinct protective coating before being placed in a capsule. This means:
- Ingredients with competing absorption pathways are physically separated
- Each beadlet can be engineered for timed or targeted release in the digestive tract
- Moisture-sensitive or volatile compounds are protected until they reach the optimal absorption site
- The interaction profile of the overall formula is controlled by design, not left to chance
This is the delivery technology at the foundation of the Art of You formula — and it explains why the discussion of pharmaceutical grade supplements cannot stop at purity. The science of delivery is inseparable from the science of absorption.
What Art of You does differently
Art of You was built on the premise that the supplement industry's structural compromise — food-grade ingredients, one-size-fits-all formulas, opaque manufacturing — was leaving too much on the table. The formula was co-developed in collaboration with a University of Westminster research partnership, bringing academic rigour to ingredient selection, dosing rationale, and delivery architecture.
Every ingredient in the Art of You formula is sourced to pharmaceutical-grade standards — including full CoA documentation, heavy metal verification, and purity confirmation at the pharmacopoeial level. The manufacturing facility operates under pharmaceutical GMP, not food GMP.
But the differentiator is not just grade. It is the combination of:
- Pharmaceutical-grade raw materials — 99%+ purity, pharmacopoeial compliance
- Individual beadlet encapsulation — each of the 29 clinically-studied ingredients delivered in its own precisely engineered micro-bead
- Biomarker personalisation — the formula is not fixed. It is adjusted to your individual blood biomarkers, so the ingredients and doses reflect your actual nutritional status
- Quarterly reassessment — as your biomarkers change, your formula changes with them
The personalisation dimension matters here because pharmaceutical grade without personalisation is still a generic product. If your Vitamin D levels are optimal, adding more Vitamin D at pharmaceutical purity does not make it more effective for you. Precision manufacturing paired with precision dosing is what closes the loop.
To understand how personalisation works in practice, take the Art of You quiz — it is the starting point for mapping your supplement formula to your biomarker profile.
Key takeaways
- Pharmaceutical grade means 99%+ purity, verified to pharmacopoeial standards (USP, BP, Ph.Eur.) — the same benchmarks required for prescription medications
- Most supplements use food-grade ingredients, which are legal and generally safe but operate at lower purity thresholds (80–95%) with less rigorous testing requirements
- GMP certification covers the facility, not the product — it is a meaningful signal but does not guarantee individual batch quality or ingredient purity
- Third-party testing and certificates of analysis are the practical tools for verifying what is actually in a specific product and batch
- Grade and delivery form work together — pharmaceutical-grade purity delivers its full potential only when the delivery architecture ensures bioavailability
- Personalisation is the final variable — the right ingredient at the right purity at the right dose for your individual biomarker profile is where the real difference is made. Explore Art of You memberships to see what that looks like in practice.
Sources: FDA — Current Good Manufacturing Practice (CGMP) Regulations, NSF GMP Certification, Pharmaceutical Grade Definition — MedX, Pharma vs Food Grade Supplements — World Pharma Today, BSCG — GMP Certified Supplements: Proof, Limits, Myths, Pharmaceutical Grade Manufacturing for Supplements — SMP Nutra, EU GMP Guideline, European Pharmacopoeia Quality Standards — ScienceDirect
Art of You products are food supplements and are not intended to diagnose, treat, cure, or prevent any disease.
FAQ
What are pharmaceutical grade supplements? Pharmaceutical grade supplements are products manufactured to the same purity and quality standards required for prescription medications. They must contain 99% or greater of the stated active ingredient, verified against pharmacopoeial standards (USP, BP, or European Pharmacopoeia), with documented limits on contaminants, heavy metals, and impurities.
What does pharmaceutical grade mean for supplement quality? It means the ingredient has been tested and documented to a higher standard than food-grade alternatives. Purity is verified at 99%+, heavy metal content must fall within strict pharmacopoeial limits, and every batch must be traceable through a certificate of analysis. The practical effect is a more consistent, more predictable ingredient with fewer unknown compounds.
What is the difference between pharmaceutical grade vs food grade supplements? Food grade supplements are manufactured to food safety standards, not pharmaceutical ones. Food-grade ingredients typically contain 80–95% of the labelled active compound, with broader tolerances for contaminants and less rigorous batch-level testing. Pharmaceutical grade raises purity to 99%+ and requires pharmacopoeial compliance. Both are legal; the difference is consistency, purity, and testing rigour.
What does GMP certified mean for supplements? GMP (Good Manufacturing Practice) certification means a manufacturing facility has been independently audited and meets documented standards for cleanliness, process control, record-keeping, and quality management. It applies to the facility and its processes — not to individual finished products. A GMP-certified facility may still produce batches that fail quality testing, which is why third-party product testing and certificates of analysis provide a more complete picture.
What should I look for on a supplement label to know it is pharmaceutical grade? Look for: explicit reference to pharmaceutical-grade or pharmacopoeial-standard ingredients (USP, BP, Ph.Eur.); a certificate of analysis available on request; third-party testing verification from NSF International, USP Verified, or Informed Sport; and manufacturing in a pharmaceutical GMP facility. These signals together indicate a higher standard of manufacturing than food-grade alternatives.
Are pharmaceutical grade vitamins better than standard vitamins? Pharmaceutical grade vitamins offer higher verified purity and more rigorous batch documentation. Whether that translates to better outcomes for you depends on a second factor: the delivery form. How a vitamin is encapsulated and released in your body affects how much you actually absorb. Pharmaceutical grade purity paired with an optimised delivery form — such as individual beadlet encapsulation — supports better bioavailability than either factor alone.
What is a certificate of analysis (CoA) for supplements? A certificate of analysis is a batch-level document from an independent laboratory that verifies the identity, purity, potency, and contaminant profile of an ingredient or finished product. It confirms that what is on the label matches what is in the product, and that heavy metals and other impurities fall within specified limits. Reputable supplement brands make CoAs available on request for each production batch.
Does Art of You use pharmaceutical grade ingredients? Yes. Every ingredient in the Art of You formula is sourced to pharmaceutical-grade standards, with full certificate of analysis documentation and heavy metal verification at the pharmacopoeial level. The manufacturing facility operates under pharmaceutical GMP. The formula also uses individual beadlet encapsulation technology to optimise the delivery and bioavailability of each of the 29 clinically-studied ingredients.
How does personalisation relate to pharmaceutical grade supplement quality? Pharmaceutical grade manufacturing ensures the ingredient is pure and consistent. Personalisation ensures it is the right ingredient at the right dose for your individual biology. A pharmaceutical-grade supplement formula adjusted to your blood biomarkers — rather than a fixed one-size-fits-all formula — combines manufacturing precision with nutritional precision. Both dimensions matter for your body to benefit from what is in the capsule.

